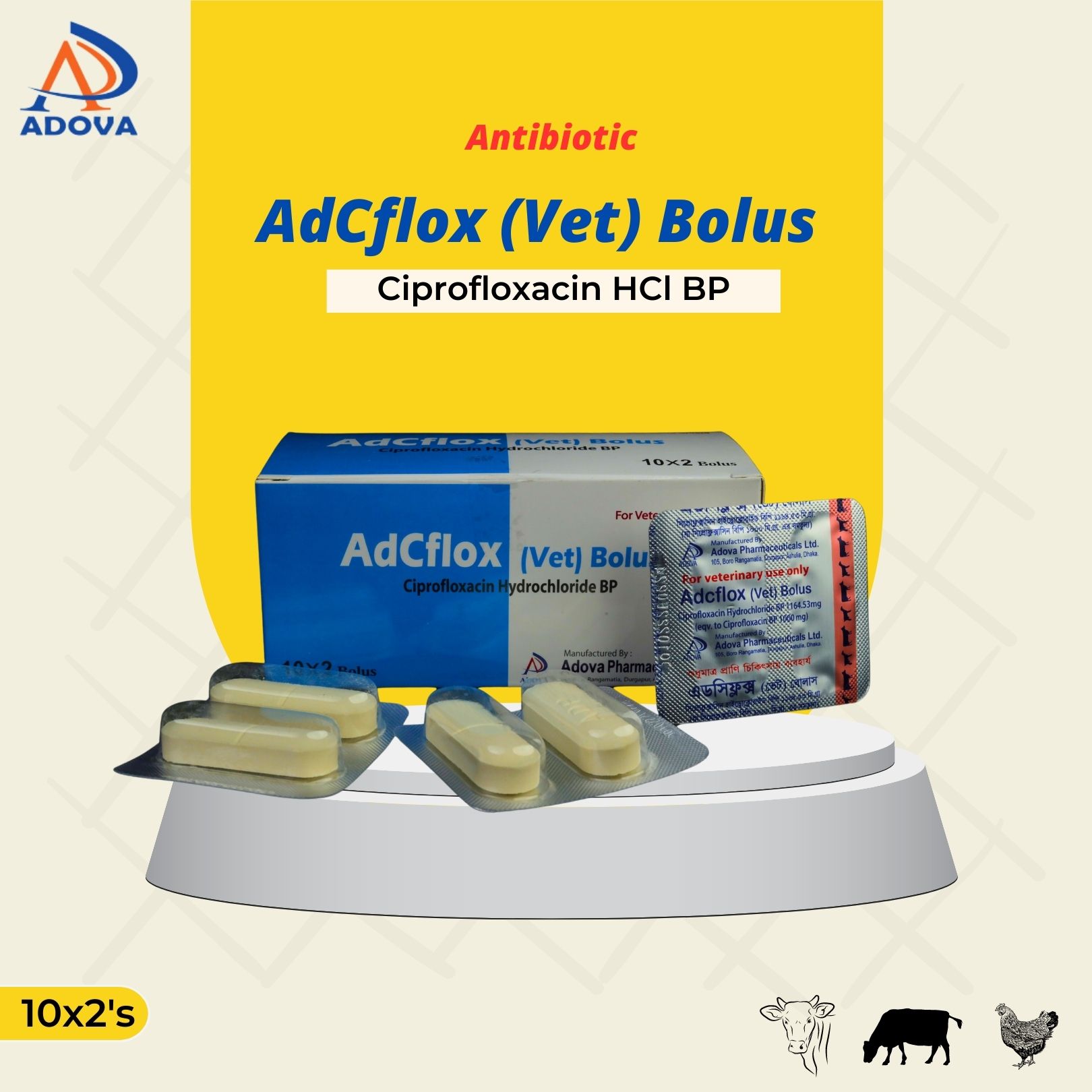
Dosage & Administration:
Cattle: 1-2 Bolus/ 70-150 kg body weight or 6.7-13.4 mg/kg body weight twice daily for 3-5 days.
Or, as directed by registered veterinary physician.
Adverse effects:
Central nervous system (CNS) toxicity; gastrointestinal reactions; hematuria; hepatotoxicity; hypersensitivity reactions; Stevens-Johnson Syndrome (blistering, itching, loosening, peeling, or redness of skin; diarrhea).
Drug Interaction:
Ciprofloxacin administered together with Theophylline can lead to elevated blood levels of Theophylline.
Precaution:
Patients receiving Ciprofloxacin should be well hydrated and should be instructed to drink fluids liberally. Because of the risk of crystalluria, it is recommended that the usual dosage of the drug should not be exceeded. Patients with a history of hypersensitivity to Ciprofloxacin or to other quinolones.
Use in Pregnancy and Lactation:
Intravenous doses of ciprofloxacin of up to 20 mg per kg of body weight (mg/kg) in pregnant rats and mice have not shown evidence of maternal toxicity, embryo toxicity, or teratogenic effects. Ciprofloxacin can be distributed into milk, sometimes at a higher concentration than in plasma, but it is not known under what conditions significant amounts might be absorbed by nursing animals.
Withdrawal Period Meat: 14 days, Milk: 3 days.









Reviews
There are no reviews yet.